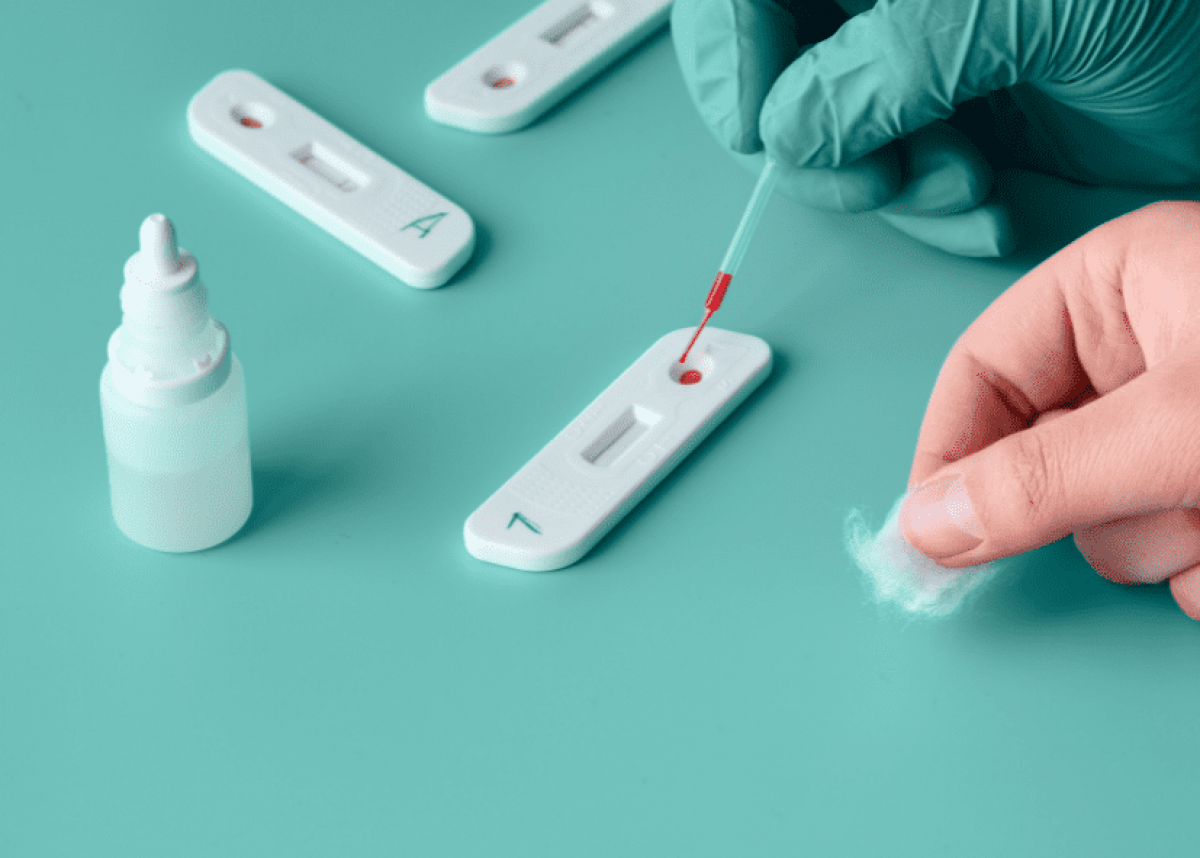
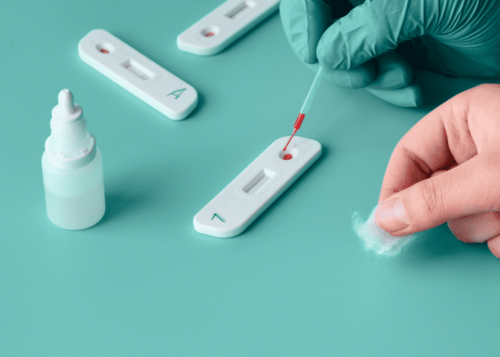
The COVID-19 virus known as SARS-CoV-2 that enters the body can be detected by the body's immune system and causes the immune system to produce antibodies to destroy it. The presence of antibodies to eradicate the COVID-19 virus can be detected through a rapid antibody test. Currently available ACCU‐TELL® COVID‐19 IgG/IgM Cassette (Whole Blood/Serum/Plasma) which has been certified by CE. This product is a rapid immunochromatographic assay that uses a combination of colored particles coated with the SARS-CoV-2 antigen to detect IgG and IgM antibodies to SARS-CoV-2 in whole blood, serum or plasma. This product can detect primary and secondary infection of SARS-CoV-2. Product packaging per box contains 25 tests, 25 pipettes, and 1 bottle of buffer solution.
This test consists of two components, namely immunoglobulin G (IgG) and immunoglobulin M (IgM). In the IgG component, anti-human IgG is coated in the IgG test strip region. During the test, the specimen reacts with the SARS-CoV-2 antigen-coated particles on the test cassette. The mixture then migrates upward on the membrane chromatographically by capillary action and reacts with anti-human IgG in the IgG test line region. If the specimen contains IgG antibodies against SARS-CoV-2, a colored line will appear on the line of the IgG test region. Likewise for the IgM component. These results indicate an antibody reaction to the presence of an antigen (reactive result).
If the specimen does not contain SARS-CoV-2 antibodies, no colored line appears indicating a non-reactive result. A colored line will always appear in the control line region (C), indicating that the specimen and buffer volumes are correct and the results can be assessed.
Features and Benefits of Rapid Test Antibody brand ACCU‐TELL® COVID‐19 IgG/IgM Cassette (Whole Blood/Serum/Plasma)
Feature
- Can use 3 types of blood samples, namely whole blood, serum, and plasma.
- The volume of the sample taken is 10 μl (little).
- The principle of the tool is an immunochromatographic assay.
- 1 bottle of buffer solution is available for 25 tests. (Additional buffer solution bottles available if needed).
- Results can be interpreted in 10 minutes (short).
- The product has the Ministry of Health's AKL permit (20303025890) and certification from CE.
Benefit
- User can select blood sample according to availability.
- Makes it easier for operators to get blood samples even in difficult/less cooperative patient conditions (young children, the elderly, people who are afraid of needles).
- Make it easy for users to read the results through the colored lines that are formed.
- Practical for use by 1 user in the laboratory.
- The user can determine the next immediate action according to the results that appear.
- Guaranteed quality according to domestic and global recommendations.
- Users can keep a certain amount of stock without worrying about the product quality being reduced.
How to use the ACCU‐TELL® COVID‐19 rapid test antibody brand IgG/IgM Cassette (Whole Blood/Serum/Plasma)
- Take a sample (whole blood, serum, plasma) according to the line on the straw and drop it on the S section of the test cassette
- 2 drops of buffer are dripped on part B of the test cassette
- Let stand for 10 minutes and interpret the results according to the image below. Do not read the results after 20 minutes.
- Reactive results in the image below are marked with numbers 2, 3, and 4. Number 2 means suspected primary infection, 3 and 4 suspected secondary infection with SARS-CoV-2. Number 1 means the result is not reactive to SARS-CoV-2.
Please note that the use of the test cassette is a maximum of 1 hour after being removed from the seal.
The antibody rapid test results should be read as reactive and unreactive. However, society in general interprets reactive results as positive and non-reactive as negative. Positive or negative results are more accurately interpreted for the diagnosis of COVID-19 with the target material for detection is the genetic material of the virus. Meanwhile, the terms reactive and unreactive are the body's immune response to incoming foreign objects (SARS-CoV-2) as the cause of COVID-19 disease.
Reference:
- Instruction for Use ACCU-TELL® COVID-19 IgG/IgM Cassette (Whole Blood/Serum/Plasma). 2020 by AccuBiotech Co., Ltd.